A direct comparison of HypoThermosol® (Biolife Solutions) and ROKEPIE (Sulfateq) was recently published in PlosONE. Freitas Robero from the group of Prof Rui Reis (CEO of the European Institute of Excellence on Tissue Engineering and Regenerative Medicine) used both agents for their intended application: The hypothermic preservation of cells for multiple days. The authors found that ROKEPIE best safeguarded the differentiation characteristics of the cells. If we include the prices of both agents, we think ROKEPIE is clearly favored at 40% of the price of HypoThermosol®.
The authors found that both agents were proficient in their intended goal of safeguarding cell viability but that only ROKEPIE was able to preserve the differentiation characteristics of human adipose tissue derived stem cells (hADSC). You can read the full article on the website of PlosONE by clicking here.
Advantages of ROKEPIE
An alternative to cryopreservation
The authors describe the need of an alternative to cryopreservation and propose pausing cells at hypothermic temperatures as a simplified alternative (Freitas-Ribeiro et al., 2019). Compared to cryopreservation, hypothermic preservation has no risk of ice nucleation and omits the need for toxic cryoprotectants. However, hypothermic preservation is known to have its own deleterious effects and may lead to cell injury and death when done for several days.
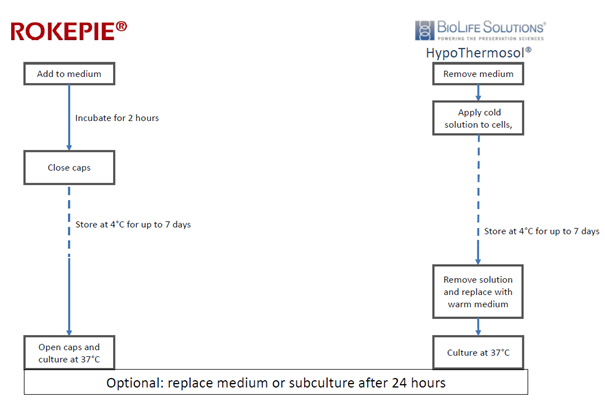
The ingredients
Under investigation are HypoThermosol (HTS) and ROKEPIE. HTS is a solution designed to balance the intracellular state at low temperatures and replaces the cell culture medium completely. The solution includes free radical scavengers, pH buffering agents, energy substrates and features osmotic support and a ionic composition that supports cells during hypothermia (Ostrowska, Gu, Bode, & Van Buskirk, 2009). ROKEPIE on the other hand is a supplement to cell culture medium. Its active ingredient is a single 6-chromanol derivative (SUL-109) that increases the activity of complex I and IV of the mitochondrial respiration chain (Hajmousa et al., 2017), thereby increasing ATP production and minimizing the production of reactive oxygen species .
The comparison
The authors compare the 2 preservation strategies and test their capacity to support the preservation of monolayers of hADSCs at 4°C for up to 7 days. The readouts included morphology, viability, phenotype, protein expression and extracellular matrix (ECM) integrity. In addition, the differentiation potential of hADSC (their capability to differentiate into adipogenic or osteogenic cells) was evaluated.
With regards to the metabolic activity of the cells, confluent hADSC preserved in HTS for 3 days showed improved metabolism (~60% of 37°C control) compared to ROKEPIE or culture medium (~40%). After 7 days, confluent cells preserved in either HTS or ROKEPIE showed improved metabolism (~25%) compared to cells preserved in culture medium (~10%). When non-confluent cells were used, only ROKEPIE was shown to improve metabolic activity or the level of double-strand DNA as a marker for cell content after 7 days. The authors found that both HTS and ROKEPIE were able to safeguard the morphology and integrity of the ECM. When the cells were tested for their capability to differentiate into adipogenic cells, the authors concluded that only cells preserved with ROKEPIE were able to differentiate properly.
Optimal concentration
ROKEPIE should be tested for your cell lines of choice at 1:20 to 1:1000 dilutions. For hADSC, the optimal concentration was found to be 10 µM, corresponding to a 1:100 dilution of the product (Hajmousa et al., 2017) . At these concentrations, 200 mL of storage medium would cost €150. In comparison, 200 mL of HTS would cost at least €372,-, making the product almost 2.5x more expensive (this calculation uses the price of a 500 mL bottle of HTS as stated on the BioLife Solutions website accessed on October 28th 2019). An added benefit of ROKEPIE is that it does not need to be replaced immediately after preservation: The agent supports cell growth at 37°C and is non-toxic. This in contrast to HTS, which needs to be replaced shortly after preservation in a phase where cells are in a particularly vulnerable state, recovering from hypothermia and rewarming.
References
Freitas-Ribeiro, S., Carvalho, A. F., Costa, M., Cerqueira, M. T., Marques, A. P., Reis, R. L., & Pirraco, R. P. (2019). Strategies for the hypothermic preservation of cell sheets of human adipose stem cells. PLOS ONE, 14(10), 1–16. https://doi.org/10.1371/journal.pone.0222597
Hajmousa, G., Vogelaar, P., Brouwer, L. A., van der Graaf, A. C., Henning, R. H., & Krenning, G. (2017). The 6-chromanol derivate SUL-109 enables prolonged hypothermic storage of adipose tissue-derived stem cells. Biomaterials, 119, 43–52. https://doi.org/10.1016/j.biomaterials.2016.12.008
Ostrowska, A., Gu, K., Bode, D. C., & Van Buskirk, R. G. (2009). Hypothermic storage of isolated human hepatocytes: a comparison between University of Wisconsin solution and a hypothermosol platform. Arch Toxicol, 83(5), 493–502. https://doi.org/10.1007/s00204-009-0419-x
